Risk Assessment & Requirements Management Made Simple
Manage risks, requirements, tests, and test results in one connected system. Purpose-built for medical device design control with ISO 14971 risk assessment built in. Enterprise traceability without enterprise complexity.
Up and running in minutes — no lengthy implementation, no complex setup.
Built for medical device design control
Purpose-built for risk management, requirements traceability, and regulatory submissions
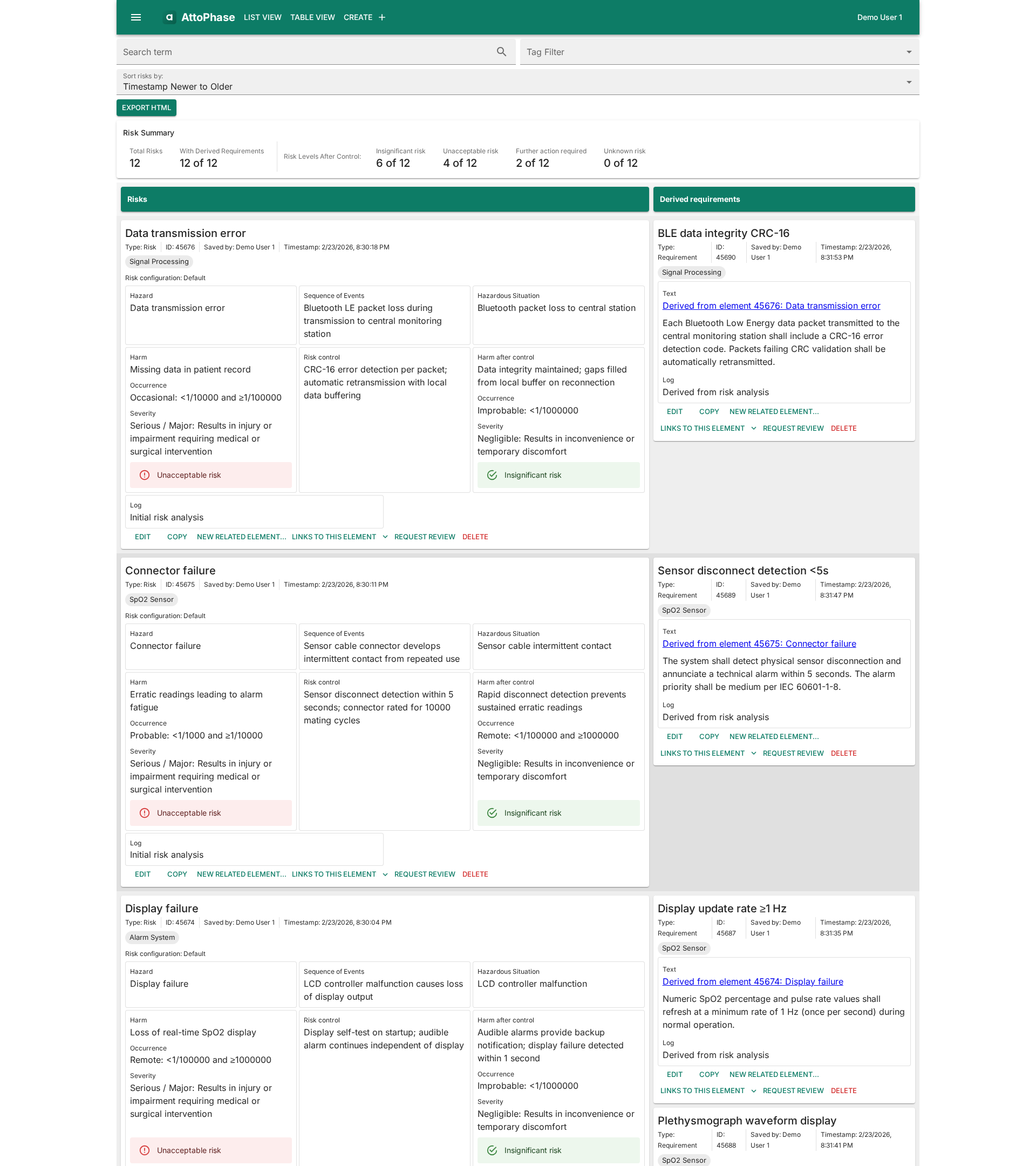
ISO 14971 Risk Assessment
Standard-compliant risk management out of the box with sensible defaults. Fully configurable severity and occurrence scales, hazard categories, and risk columns — adapt the process to your needs without losing compliance.
Requirements Management
Never miss a requirement. Track design inputs and outputs with full version history, and link every requirement to its tests, risks, and related elements. Simple, flexible traceability matrices make it easy to see the full picture.
Import/Export & Zero Lock-in
Import existing requirements and risks from Excel. Export your data to Excel or to print-ready HTML for Word and PDF. AI-ready open formats — your data stays yours, no lock-in.
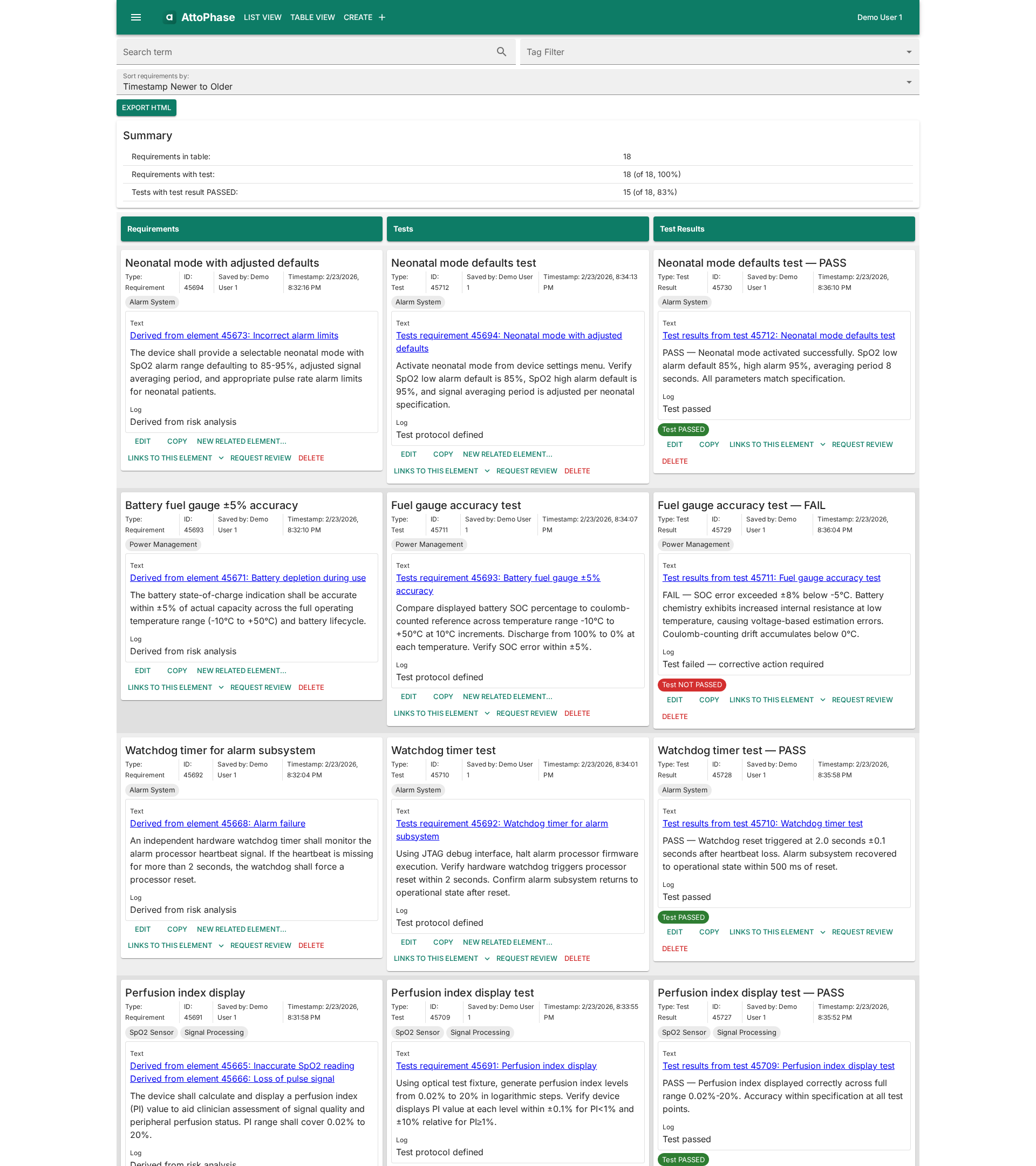
Full Traceability
Trace from user needs through design requirements, verification, and validation. Dedicated traceability tables show requirement-test-result and risk-requirement mappings at a glance.
Test Management
Link verification and validation tests to requirements and record results with pass/fail status. See at a glance which requirements are fully verified.
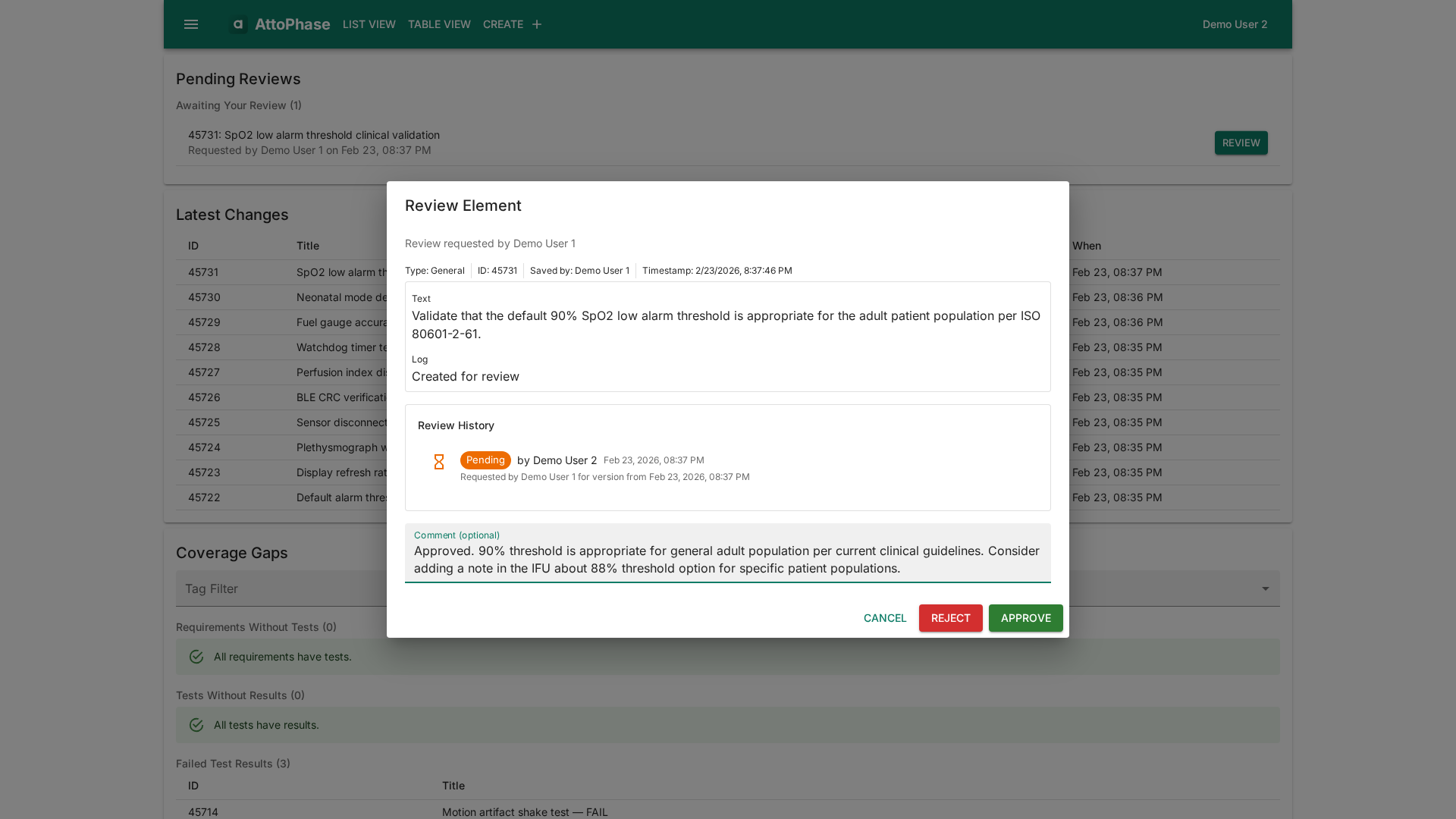
Review & Approval Workflows
Request and track reviews on any element. Keep an audit trail of who reviewed what and when — built for design control and regulated approval processes.
Document Attachments
Attach supporting documents to any element. Keep test evidence, design specifications, and references organized alongside your requirements.
Flexible Tagging
Use tags to organize by product, device variant, project phase, or any custom categorization. Filter and search across all element types with ease.
Team Collaboration
Organize work in lockers with granular permissions. Control who can view, edit, or administer each area across your organization.
Simple, transparent pricing
Choose the plan that fits your team. Request access to get started.
Basic
For small teams getting started with risk and requirements management
- Up to 500 elements
- 5 users included, then €25/month each
- ISO 14971 risk assessment
- Requirements management
- Full traceability tables
- Excel & HTML import/export (PDF/Word-ready)
- Review workflows
- Email support
Plus
For growing teams that need document storage and more capacity
- Everything in Basic
- Up to 5,000 elements
- 20 users included, then €25/month each
- Document attachments with 10 GB storage
- Priority support
Custom
For organizations that need tailored limits or features
- Tailored element, storage, and user limits
- All features included
- SSO integration
- Dedicated support
- Custom onboarding
Request Access
Interested in AttoPhase? Fill out the form below and we'll be in touch to set up your account.
Or email us directly at hello@attophase.com